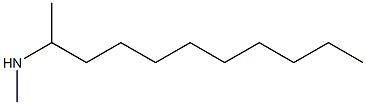
Alcohols, C>10, beta-(methylamino)-
Alcohols, C>10, beta-(methylamino)-
CAS: 68439-58-7
Molecular Formula: C12H27N
Alcohols, C>10, beta-(methylamino)- - Names and Identifiers
| Name | Alcohols, C>10, beta-(methylamino)- |
| Synonyms | Einecs 270-408-3 |
| CAS | 68439-58-7 |
| EINECS | 270-408-3 |
Alcohols, C>10, beta-(methylamino)- - Physico-chemical Properties
| Molecular Formula | C12H27N |
| Molar Mass | 185.34948 |
Alcohols, C>10, beta-(methylamino)- - Introduction
Alcohols, C>10, beta-(methylamino) is a class of organic compounds containing hydroxyl groups in the molecule, the number of carbon atoms is greater than 10, and there is a methyl amino group in the beta position of the molecular structure.
The properties of such alcohol compounds will vary depending on the specific molecular structure. As an organic compound, they are usually colorless liquids or solids with a specific odor at room temperature. They are readily soluble in polar solvents such as water and alcoholic solvents.
This type of alcohol has a wide range of uses in practical applications. One of the main uses is as a solvent for dissolving and mixing other substances in the chemical and industrial fields. They can also be used in the synthesis of other compounds such as fragrances, plant extracts and pharmaceutical intermediates.
There are a variety of methods for preparing such alcohols. A common method is to synthesize them by carbon-hydrogen bond insertion reactions or hydroxylation reactions. The specific preparation method can be adjusted according to different molecular structures, and needs to be selected according to specific conditions.
In terms of safety information, such alcohol compounds are generally considered to be relatively safe. However, as with all chemicals, proper handling and safety measures need to be observed. These compounds may have certain irritants, such as eye and skin irritation. When using these compounds, care must be taken to avoid skin contact and inhalation. At the same time, these compounds must be stored and handled properly to avoid dangerous situations such as fire and explosion.
Summary: Alcohols, C>10, beta-(methylamino) is a class of organic compounds with a wide range of applications, with specific properties, can be used in solvents, synthesis of other compounds and other fields. Their preparation methods are varied and require attention to safety measures during use.
The properties of such alcohol compounds will vary depending on the specific molecular structure. As an organic compound, they are usually colorless liquids or solids with a specific odor at room temperature. They are readily soluble in polar solvents such as water and alcoholic solvents.
This type of alcohol has a wide range of uses in practical applications. One of the main uses is as a solvent for dissolving and mixing other substances in the chemical and industrial fields. They can also be used in the synthesis of other compounds such as fragrances, plant extracts and pharmaceutical intermediates.
There are a variety of methods for preparing such alcohols. A common method is to synthesize them by carbon-hydrogen bond insertion reactions or hydroxylation reactions. The specific preparation method can be adjusted according to different molecular structures, and needs to be selected according to specific conditions.
In terms of safety information, such alcohol compounds are generally considered to be relatively safe. However, as with all chemicals, proper handling and safety measures need to be observed. These compounds may have certain irritants, such as eye and skin irritation. When using these compounds, care must be taken to avoid skin contact and inhalation. At the same time, these compounds must be stored and handled properly to avoid dangerous situations such as fire and explosion.
Summary: Alcohols, C>10, beta-(methylamino) is a class of organic compounds with a wide range of applications, with specific properties, can be used in solvents, synthesis of other compounds and other fields. Their preparation methods are varied and require attention to safety measures during use.
Last Update:2024-04-09 21:53:21